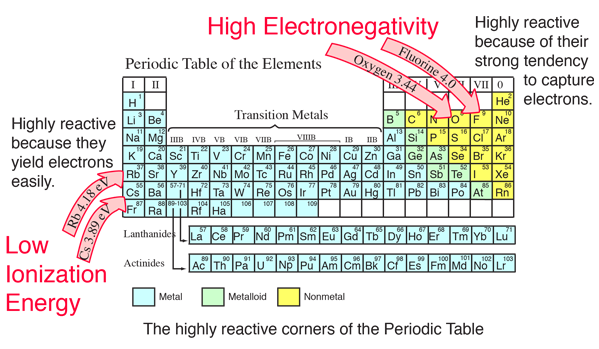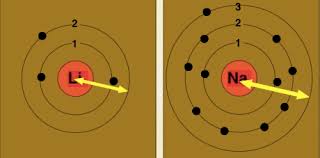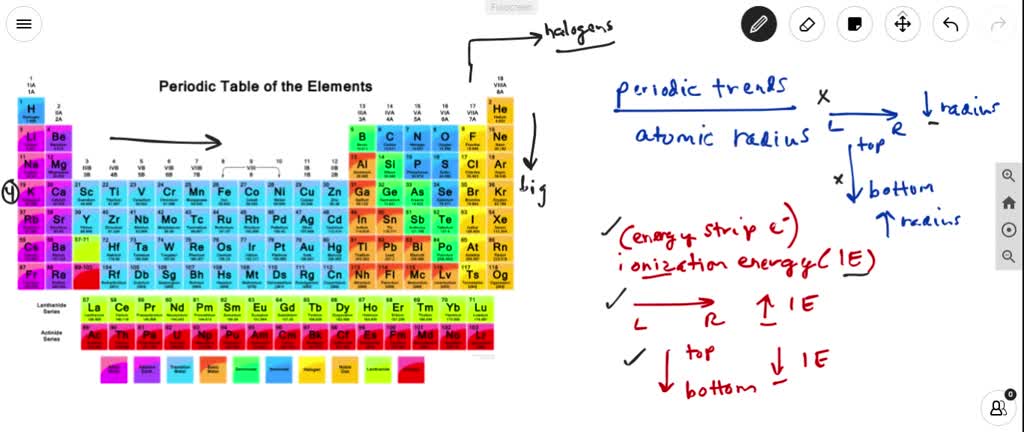
SOLVED:(a) What is the trend in first ionization energies as one proceeds down the group 17 elements? Explain how this trend relates to the variation in atomic radii. (b) What is the

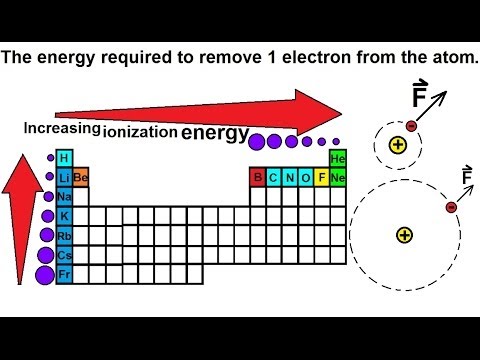
Ionization Energy: Periodic Table Trends | What is Ionization Energy? - Video & Lesson Transcript | Study.com

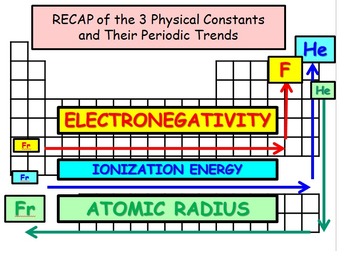
Periodic Trends: Electronegativity; Ionization Energy; and Atomic Radius | Teaching chemistry, Ionization energy, Chemistry lessons
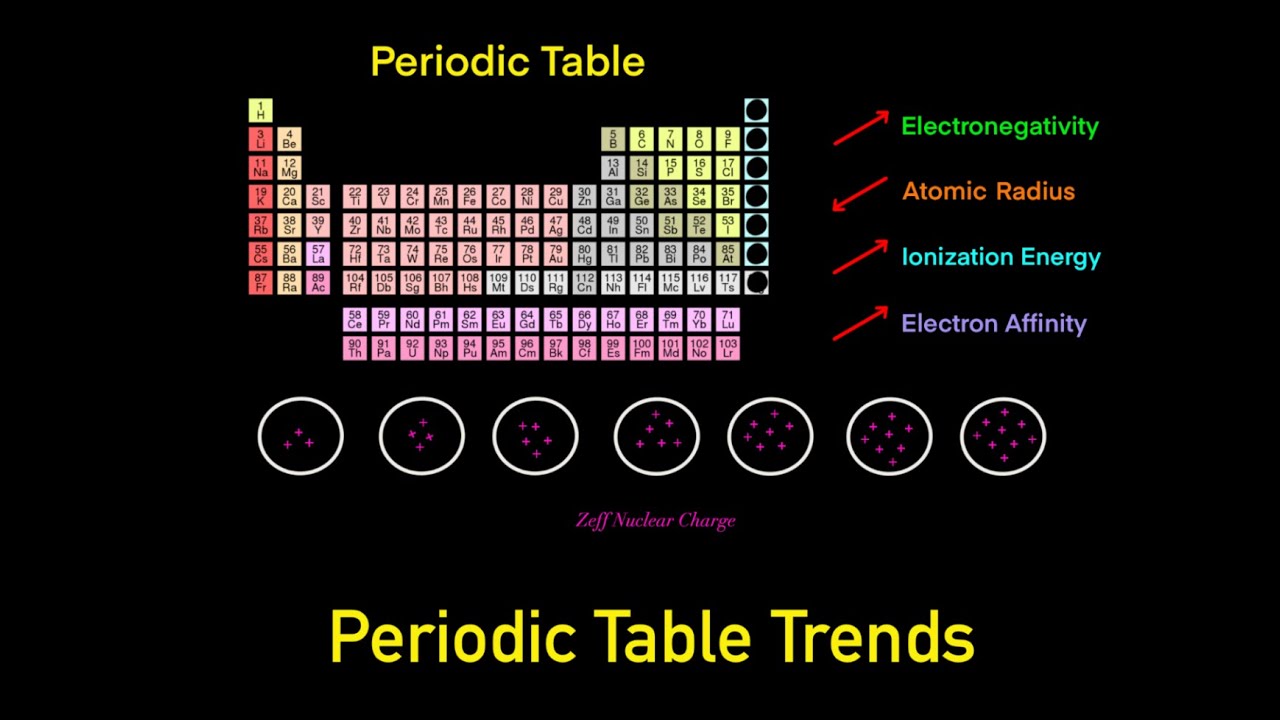
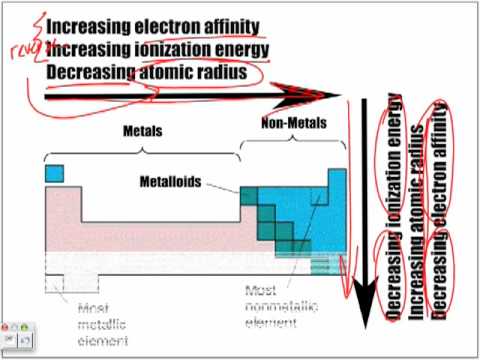
Periodic Table Trends Trick (Electronegativity, Atomic Radius, Ionization Energy, Electron Affinity) - YouTube

Department of Chemistry,University of Kashmir - Chart of Periodic Table Trends... Use this chart to see at a glance the periodic table trends of electronegativity, ionization energy, atomic radius, metallic character, and
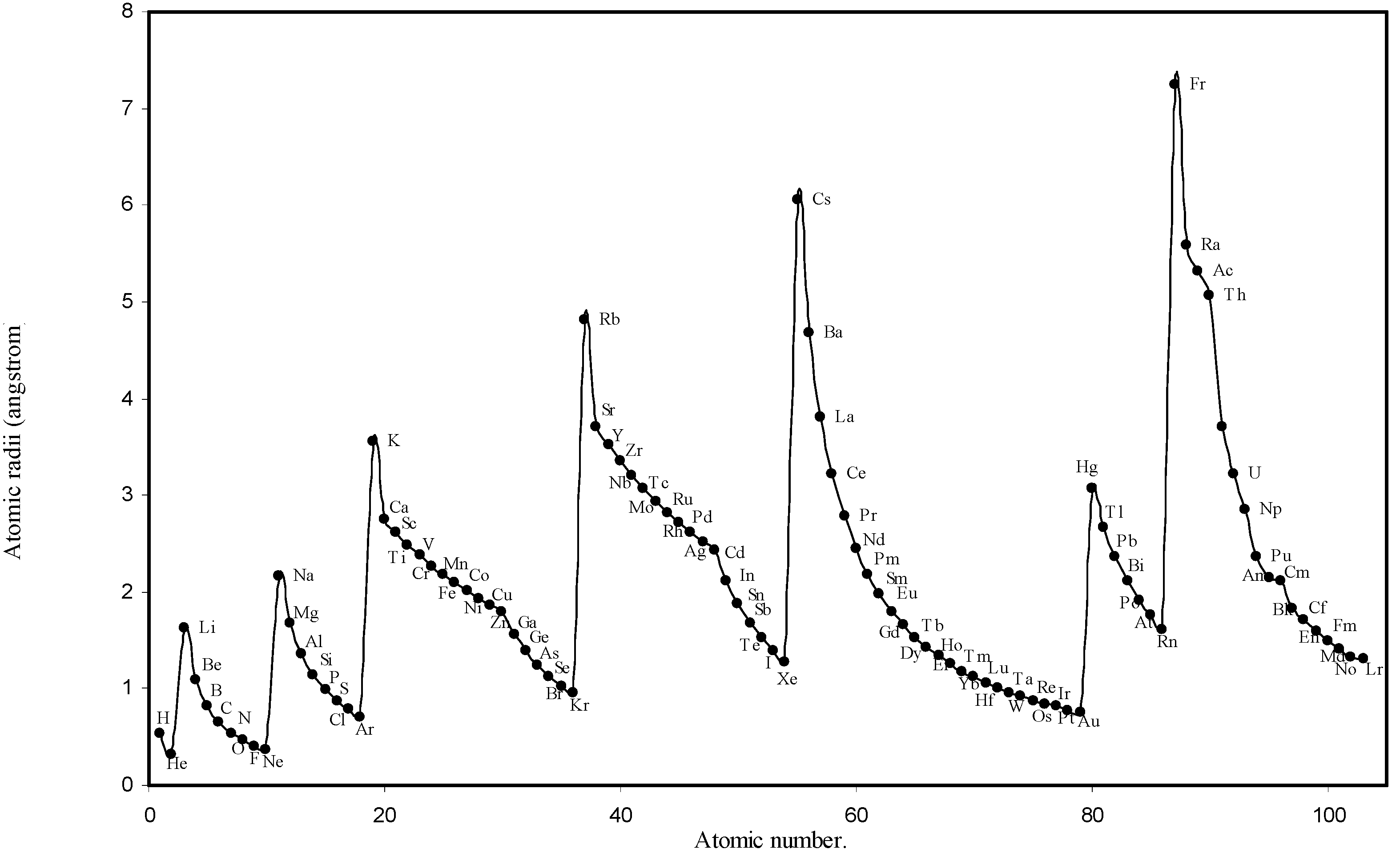
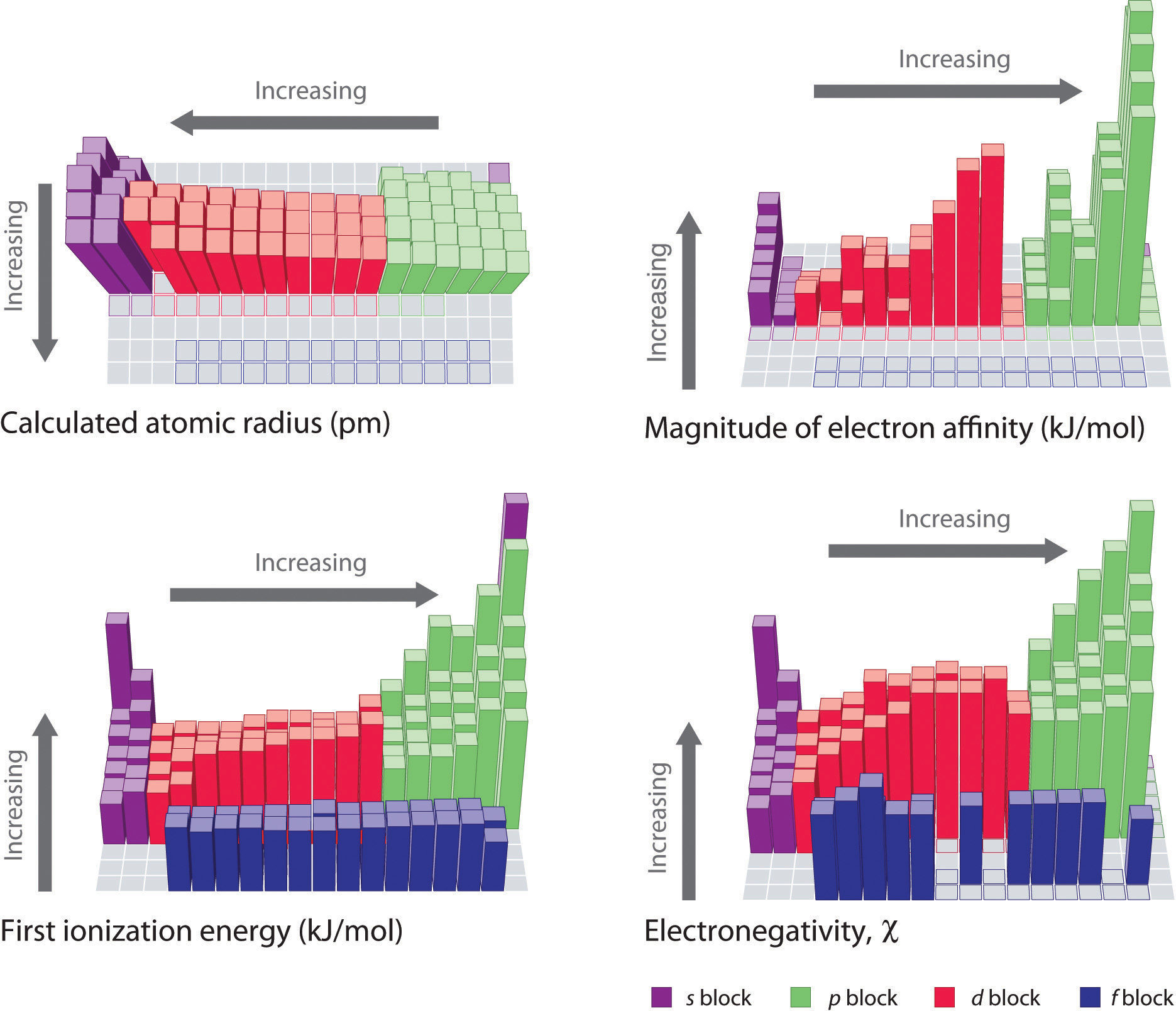
IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 1. The Atomic Radii